Maintaining a Healthy Environment for Your Water Pets
Fishkeeping as a hobby? Why not! After all, who doesn’t get mesmerized just by the view of live plants and fish swimming gracefully in clean and clear water. That is why keeping your aquarium clean at all times is important to the wellbeing of your aquatic pets; and to keep your fish healthy and happy, you will need to do a few regular maintenance jobs.
Daily maintenance
- Turn on lights on scheduled intervals within the day (use of a timer is recommended).
- Feed your fish once or twice a day, making sure they consume what you feed them within 1 to 2 minutes.
Weekly maintenance
- Clean the inner surface of your aquarium; and make sure to rid of algae and other wastes that have settled at the bottom.
- Replace water by approximately 10% to 15%. A gravel vacuum can be used to vacuum the bottom of the aquarium and remove excess organic matters.
- Always condition the water for replacement to approximately the same temperature as that in the aquarium to avoid shocking your fish.
- Depending on the size of our aquarium, add biological complexes whenever you change your aquarium water to stimulate the biological action of the ecosystem and keep the water in optimal conditions.
- Prune the plants as necessary.
- Analyze the physical/chemical quality of the water.
Below is detailed explanation of the parameters of water quality.
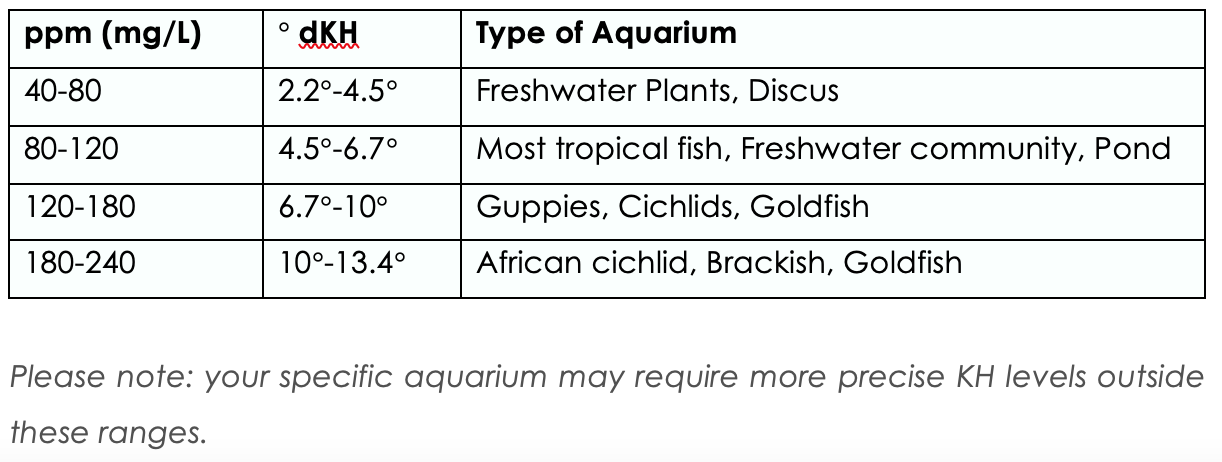
General Hardness
Hardness is a measurement of the dissolved minerals in water. Soft water contains low mineral content, while hard water contains otherwise. General hardness appears to be the critical factor for a variety of biological processes, most notably reproduction. The eggs of many egg-laying fishes only seem to develop properly within a certain general hardness range. The precise mixture of minerals in the water will vary according to the sediments through which the water has flowed. Water from a chalk aquifer, for example, will have a high concentration of calcium carbonate.
The chart below is a guide of desired GH for different types of fish.
Nitrate
Although invisible and odorless, nitrate can have a deadly effect on a tank’s inhabitants. Many fish will not show any symptom of nitrate poisoning until the level reaches 100 ppm or even higher, but studies have shown that long-term exposure to sub-critical concentrations of nitrate stresses fish, making them more susceptible to diseases, hampers growth of the young and decreases reproduction rate. Ideal Nitrate level is below 50 ppm.
Nitrite
Nitrite is a highly toxic pollutant which is produced by bacteria during the breakdown of fish wastes and other organic matters through a process called nitrification. Overfeeding and overstocking can lead to high nitrite level, but incorrect filter maintenance and new tank syndrome are perhaps the most common cause of high-level nitrite detections. In an established aquarium, nitrite level should be maintained at 0 ppm.
Free Chlorine
Free chlorine is a halogen chemical element. Concentrated levels of chlorine kill fish and other aquatic life-forms. It is an oxidizer which removes the necessary slime coat from fish, causing stress and stress-related illnesses. As the tank’s pH levels go down, chlorine becomes more toxic. Exposure to high levels of chlorine damages the gill structure of the fish which leads to difficulty in breathing causing them to swim erratically or gasp at the water surface to get enough oxygen. These symptoms can easily be mistaken for low-oxygen problems, gill parasites or other diseases when tap water is to blame. A chlorine level of 0 ppm is ideal.
Carbonate
The carbonate hardness scale is based on the concentration of carbonate and bi-carbonate and reflects the buffering capacity of the water. Water with a high buffering capacity resists changes in pH, resulting to a stable water condition which is essential for the maintenance of many species of fish that cannot tolerate even small pH changes, such as Tanganyikan cichlids. Excessively low carbonate hardness level is not good for aquarium plants, especially those that extract some of the carbon used for photosynthesis from the carbonate and bicarbonate salts in the water.
Below is a guide of desired GH for different types of fish.
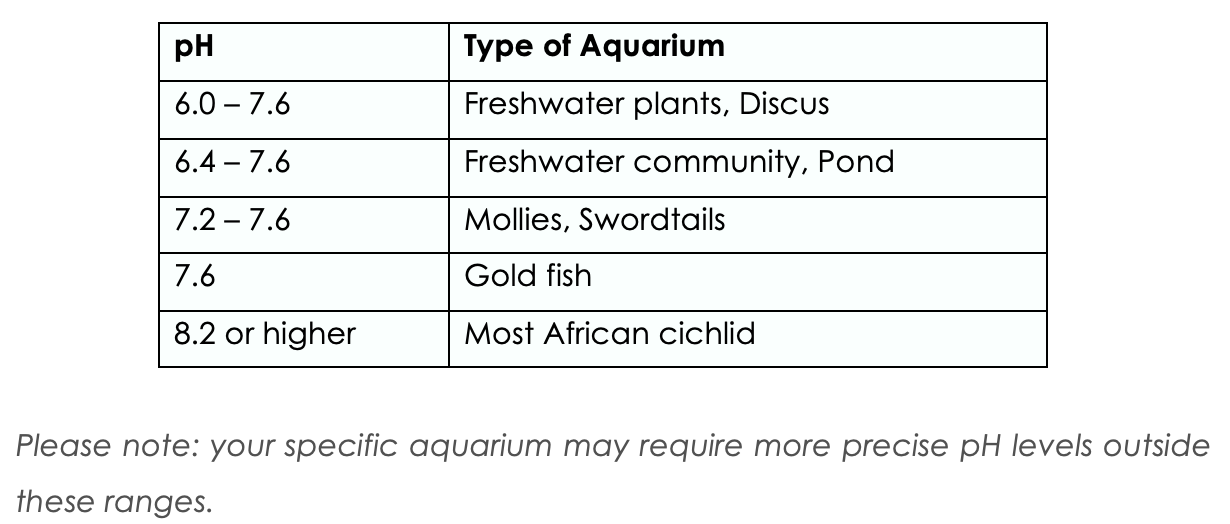
pH
The measure of the hydrogen ion(acid) concentration in water is called pH. This is the measurement of the acidity or alkalinity of water. Values above 7 are alkaline or basic; those below 7 are acidic. The scale runs from 0-14, with 0 being the most acidic and14 being the most alkaline or basic. Pure water has a pH of 7 that is said to be neutral. Most of the fish traded for hobby come from habitats where the pH lie somewhere between pH 6 and pH 8. All fish have a preferred pH range, and one of the factors every aquarist must consider is matching the conditions in the aquarium to that of their natural habitat.
The chart below is a guide on the desired GH for different types of fish.
Monthly maintenance
- Clean the filtration equipment with water from the same aquarium. Avoid using chemical products such as soap, bleach and other cleaning agents as these might contaminate the equipment and later poison the fish. Keep in mind that the frequency of filter cleaning also depends on the type of the filter we use; sponge filters need to be cleaned more frequently than canister filters.
- Charge and replace filter media. Avoid replacing all filtering charges on the same day. Instead, do it every 2 to 4 weeks to avoid the loss of biological population.
So you see, keeping your aquariums clean is not as complicated as you think. Keep a maintenance chart next to your aquarium so you don’t miss a schedule. That way, you always keep your aquariums in sparkling condition. A hazy aquarium isa stressful environment for your fish and is a total eye sore. A well-preserved, spotless, and well-lighted aquarium, with healthy live plants and fish will always be a source of good therapy and relaxation for everyone.